Hormone-Related Cancers and Stem Cell Pathobiology

Salvatore Pece
Unit Director
| [email protected] | |
| Location |
Building 13
Floor 3rd Via Adamello 16, Milano |

Our lab aims to understand the cellular and molecular basis of tumor heterogeneity in order to develop biology-driven approaches for the treatment of cancer patients. We are particularly interested in cancer stem cells (CSCs) and how they contribute to tumor heterogeneity and drive tumorigenesis, therapy failure and metastasis.
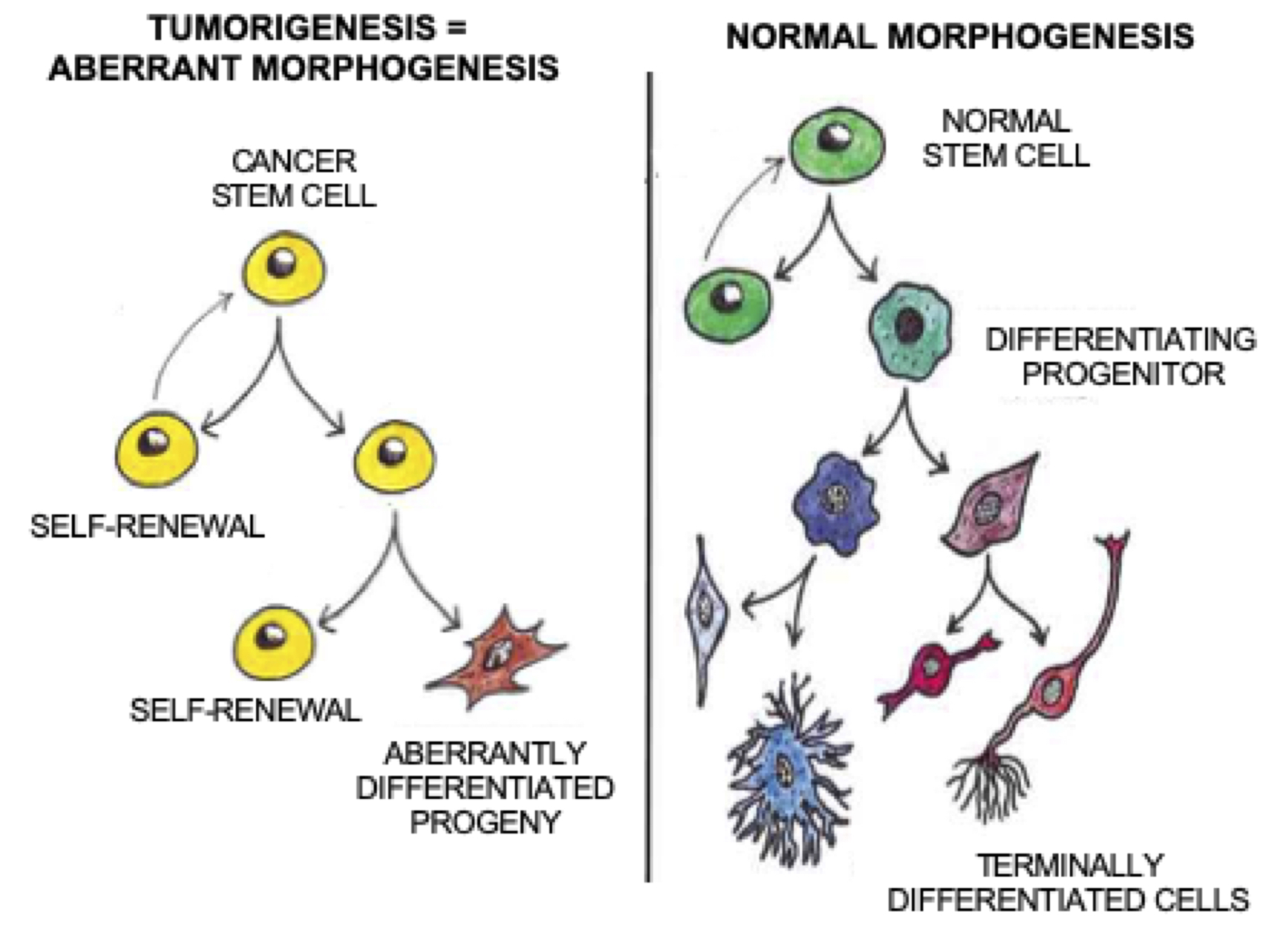
The CSC theory proposes that tumorigenesis can be viewed as normal tissue morphogenesis gone awry, with the formation of tumors being driven by stem-like cells that display abnormal self-renewal properties, resulting in the generation of aberrantly differentiated progenitors and an expanded heterogeneous CSC compartment.
Despite increasing evidence supporting the existence of CSCs, their exact identity in many cancers has remained elusive. By studying the molecular identity and pathobiology of CSCs, we aim to identify prognostic/predictive biomarkers for patient stratification and tailoring therapy, and novel molecular targets for personalized therapy.
This approach has been applied to breast cancer where we have developed a prognostic test, StemPrintER, based on the transcriptional profile of breast SCs (Pece S. 2019 EBioMedicine). We have also determined that loss of the tumor suppressor protein Numb in breast cancer is responsible for aberrant CSC self-renewal and tumorigenesis and correlates with poor prognosis. Notably, targeting Numb dysfunction represents a selective anti-CSC therapy for aggressive breast cancers (Tosoni D. 2017 EMBO Mol Med).
Most Relevant Publications
-
Salemme V, Vedelago M, Sarcinella A, Moietta F, Piccolantonio A, Moiso E, Centonze G, Manco M, Guala A, Lamolinara A, Angelini C, Morellato A, Natalini D, Calogero R, Incarnato D, Oliviero S, Conti L, Iezzi M, Tosoni D, Bertalot G, Freddi S, Tucci FA, De Sanctis F, Frusteri C, Ugel S, Bronte V, Cavallo F, Provero P, Gai M, Taverna D, Turco E, Pece S, Defilippi P
p140Cap inhibits β-Catenin in the breast cancer stem cell compartment instructing a protective anti-tumor immune response.
Nat Commun, 2023
-
Filippone MG, Freddi S, Zecchini S, Restelli S, Colaluca IN, Bertalot G, Pece S, Tosoni D, Di Fiore PP
Aberrant phosphorylation inactivates Numb in breast cancer causing expansion of the stem cell pool.
J Cell Biol, 2022
-
Filippone MG, Gaglio D, Bonfanti R, Tucci FA, Ceccacci E, Pennisi R, Bonanomi M, Jodice G, Tillhon M, Montani F, Bertalot G, Freddi S, Vecchi M, Taglialatela A, Romanenghi M, Romeo F, Bianco N, Munzone E, Sanguedolce F, Vago G, Viale G, Di Fiore PP, Minucci S, Alberghina L, Colleoni M, Veronesi P, Tosoni D, Pece S
CDK12 promotes tumorigenesis but induces vulnerability to therapies inhibiting folate one-carbon metabolism in breast cancer.
Nat Commun, 2022
-
Pece S, Sestak I, Montani F, Tillhon M, Maisonneuve P, Freddi S, Chu K, Colleoni M, Veronesi P, Disalvatore D, Viale G, Buus R, Cuzick J, Dowsett M, Di Fiore PP
Comparison of StemPrintER with Oncotype DX Recurrence Score for predicting risk of breast cancer distant recurrence after endocrine therapy.
Eur J Cancer, 2022